Our mission with Zilver PTX—and with every product we make at Cook Medical—is to try to improve patients’ lives. We have released extensive data around Zilver PTX, and this device continues to show benefits across a wide spectrum of patients.
Only Zilver PTX combines the antiproliferative drug paclitaxel with efficient, polymer-free drug delivery and vessel support to demonstrate superior 5-year results against percutaneous transluminal angioplasty (PTA) and Zilver bare-metal stents.




Please click the button below and submit the required information to connect with your local Cook representative. This form is intended for US-based physicians only.
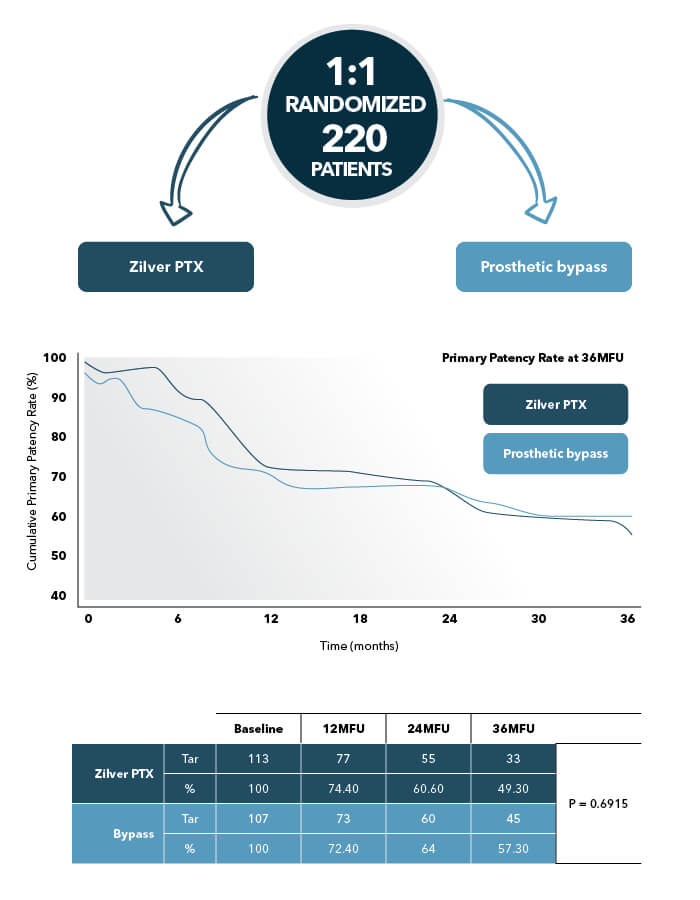
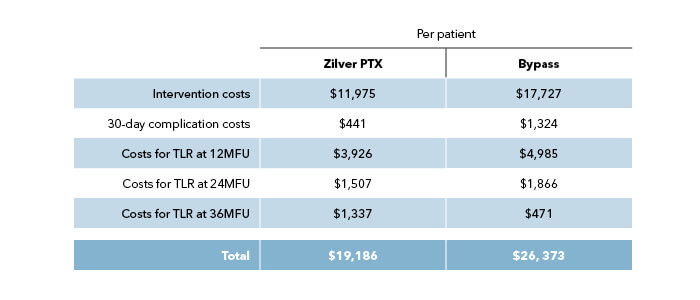
Economic analysis, taking into account procedural, hospitalization, and reintervention costs, showed a clear cost-benefit for Zilver PTX, both for the Germany and USA reimbursement models.3